Molecular geometry: The Lewis structure of CO proves that the molecule has a linear geometry, with the carbon atom and the oxygen atom both are present in a line.Electron arrangement: The Lewis structure of CO simplifies t that each oxygen atom has six valence electrons as it is of 16th group element ND, the carbon atom has four valence electrons because it represents the 14th group of periodic table, and all electrons are paired to form an stable structure. 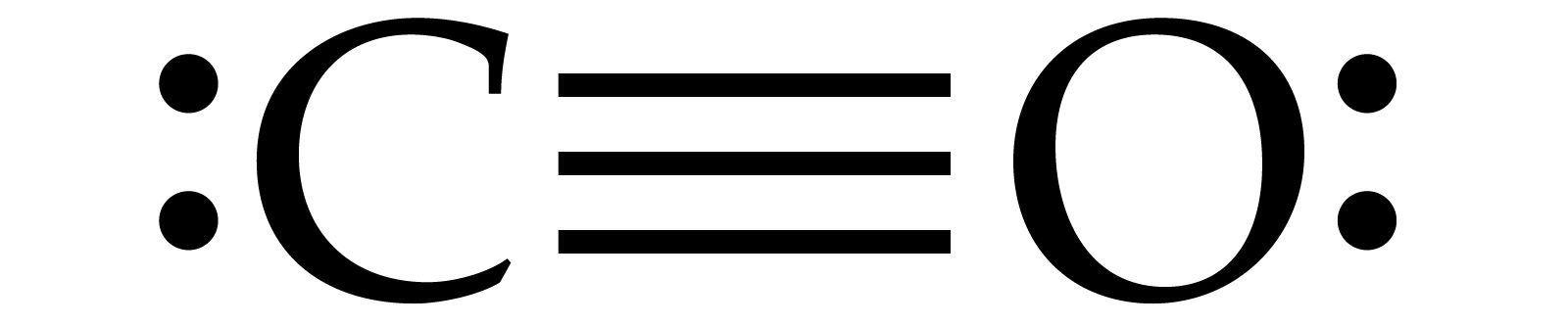 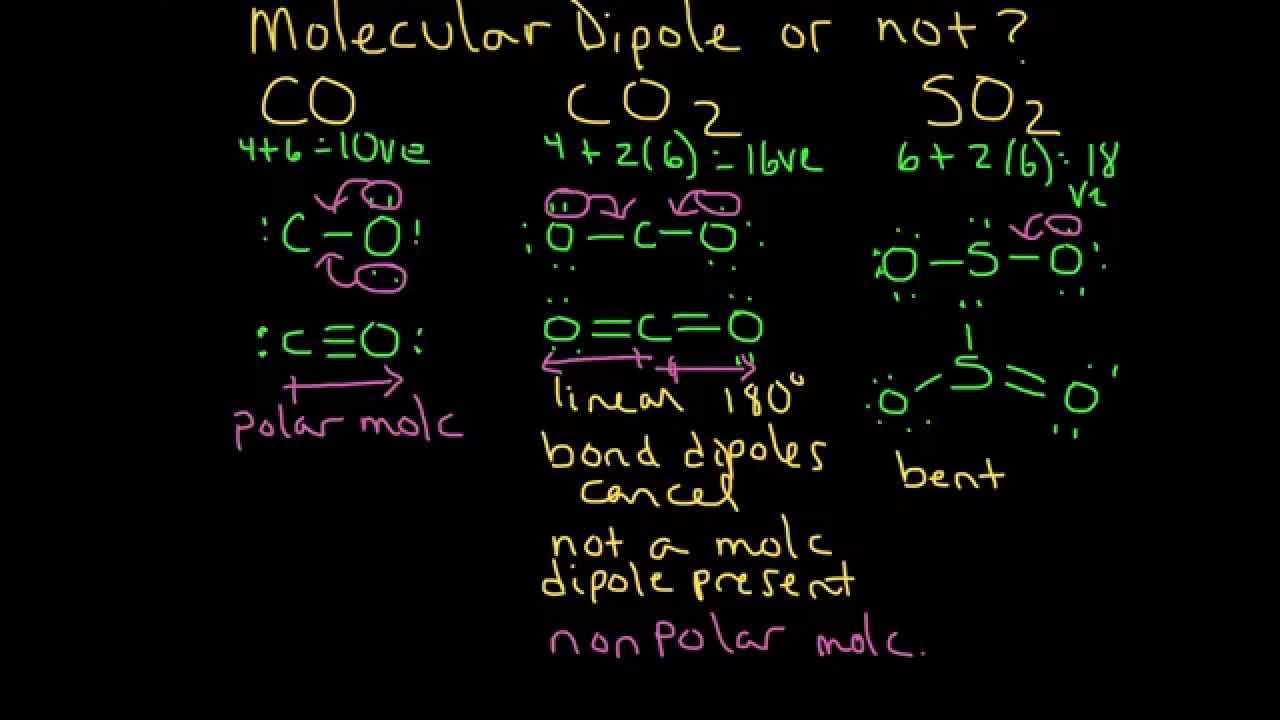
Atom arrangement: The Lewis or dot structure of CO ensures that it consists of one carbon atom and oxygen atom which are bounded by sharing of electrons and from a triple bonds between the carbon atom and oxygen atom.Here, carbon is the least electronegative atom and oxygen s more so, it will be the central atom. In the molecule of carbon monoxide (CO) it is clear fro the formula that only two atoms are there in which there will be no central atom. As carbon atom is fro 14th group then it has four valence electrons, while each oxygen atom has six valence electrons as it is of 15th gruop and the total number of valence electrons in CO is: In CO one should know the number of valance electrons for each atom in the valance shell. Step 1: Calculate the total number of electrons Steps to draw the Lewis Structure of CO (carbon monoxide) The Lewis structure of CO tells that the carbon atom has four valence electrons as it is a 14th group element, while the oxygen atom has six valence electrons though itis a 16th group elelment. The Lewis structure or a dot structure of a carbon monoxide (CO) molecule is used to represent the electronic arrangements around each atom after the atoms bounded together to form a molecule.ĬO (carbon monoxide) is a linear molecule having a carbon atom and oxygen atom bounded togather by hybridisation. Steps to draw the Lewis Structure of CO (carbon monoxide).Non-polar molecules do not have electronegativity differences between atoms and therefore do not have charge separation or a dipole moment. This charge separation results in a permanent dipole moment in polar molecules. Interpretation: We are asked to explain the reason for the small dipole moment of carbon monoxide based on the previous parts.Ĭoncept Introduction: Polar molecules have charge separation due to the difference in electronegativity between the atoms in a molecule. The formal charge on atoms in a Lewis structure is calculated using the following equation:įC = valence electrons − number of non-bonding valence electrons − 2 number of shared electrons Interpretation: We are asked to calculate the formal charge of atoms in carbon monoxide.Ĭoncept Introduction: Formal charge is the charge carried by an atom in a molecule, assuming that all chemical bonds are shared equally between the two atoms, regardless of their relative electronegativity. It represents the valence electrons of an atom using dots and shows how valence electrons are distributed within a molecule.Įxplanation: The Lewis structure of carbon monoxide is drawn as follows: Interpretation: We are asked to draw the Lewis structure for carbon monoxide.Ĭoncept Introduction: A Lewis structure is a way to show the sharing of electrons between atoms in polar covalent bonds or covalent bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
